OSM Industry Intel • Spring 2026
Liposomal Delivery for Skin Repair: What New Wound Healing Research Means for Topical Supplement Brands
A major 2026 review maps the clinical progress of liposome-based delivery systems for skin wound healing. Here is why topical supplement brands should be paying close attention.
Most topical supplements share a fundamental problem. You can put the best active ingredient in the world into a cream, gel, or lotion, and a significant portion of it will never reach the tissue where it actually needs to work. The outer layer of skin is remarkably good at keeping things out. That is literally its job. For formulators and brand owners building recovery topicals, muscle gels, or skin wellness products, this absorption barrier is the single biggest limitation standing between a product that feels nice on the surface and a product that delivers measurable results.
Liposomal delivery has been quietly solving this problem in pharmaceutical drug delivery for years. And a comprehensive new review published in Frontiers in Bioengineering and Biotechnology (Ma et al., 2026) makes a strong case that liposome-based delivery systems are now mature enough to reshape how we think about topical product development across the broader wellness and supplement space.
The review covers decades of research on liposomal systems designed specifically for skin wound healing, including infected wounds, diabetic wounds, burns, and surgical wounds. It catalogs dozens of formulations, from stimulus-responsive liposomes that release their payload in response to temperature or pH changes, to composite systems that combine liposomes with hydrogels, chitosan, and electrospun nanofiber membranes for sustained release and enhanced stability.
For supplement brands building topical product lines, this body of research offers something more valuable than abstract science. It offers a roadmap for the delivery technologies that are about to move from clinical settings into consumer wellness products. And the brands that understand these systems early will have a significant formulation advantage.
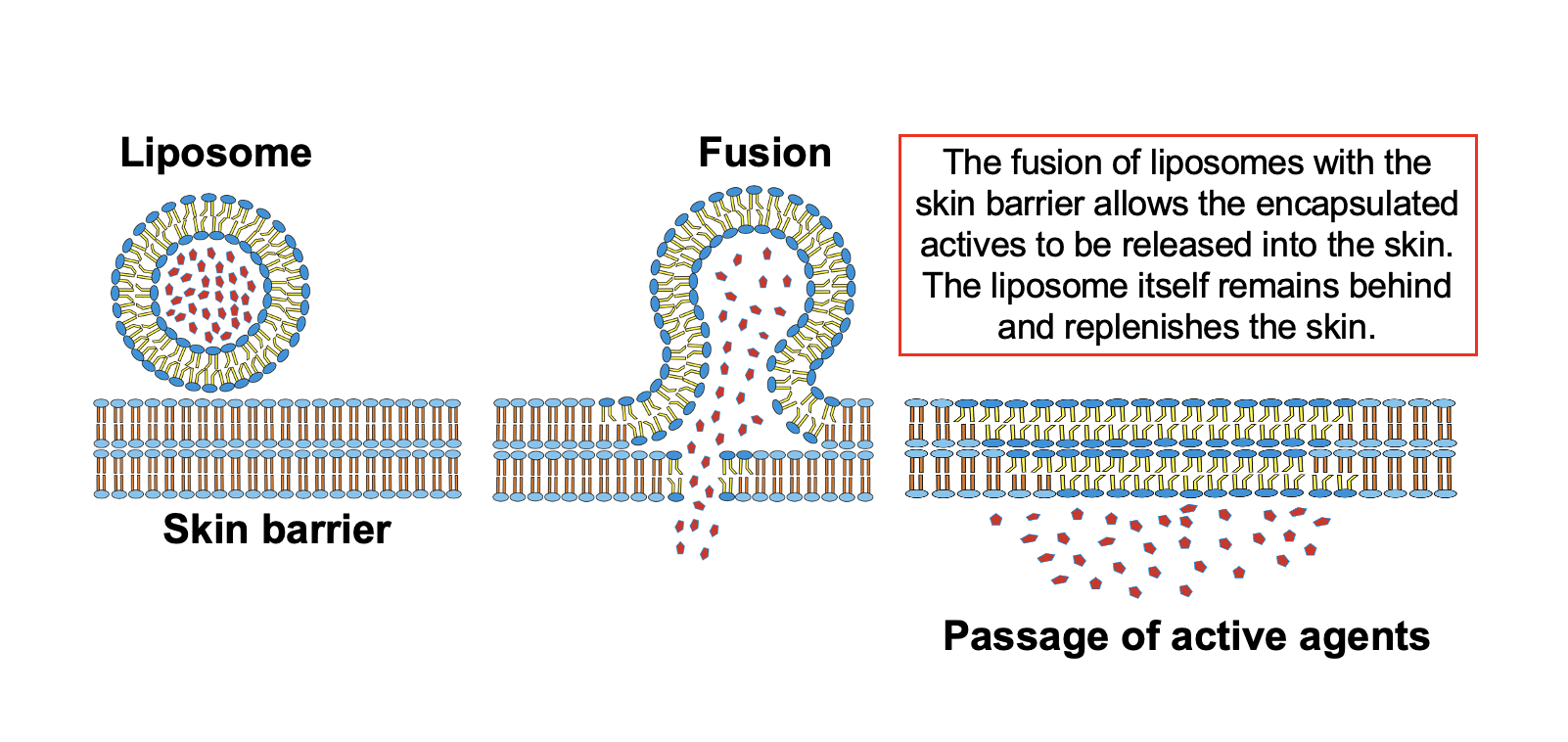
Why Liposomal Topicals Outperform Conventional Formats (and What the Clinical Data Shows)
To understand why this research matters for consumer topicals, you need to understand the core problem that liposomes solve at the skin level. As we covered in our guide to liposomal supplement technology, a liposome is a microscopic sphere made from a phospholipid bilayer, the same type of fat structure that makes up human cell membranes. Because the outer shell of a liposome is structurally identical to what the body already recognizes, these vesicles can merge with skin cells and deliver their contents directly into the tissue.
Standard topical formulations (creams, gels, ointments) deposit active ingredients on the surface of the skin and rely on passive diffusion to push them deeper. That works for some compounds, but many functional actives (curcumin, growth factors, certain peptides, herbal extracts) are either too large, too fragile, or too poorly soluble to penetrate effectively on their own. They sit on the surface, evaporate, or degrade before reaching the target tissue.
The review catalogs multiple clinical and preclinical examples where liposomal encapsulation solved these problems in measurable ways:
Curcumin delivery. Curcumin is one of the most studied anti-inflammatory compounds in topical formulation, but its poor water solubility and rapid degradation make it notoriously difficult to deliver through the skin. Multiple studies in the review show that liposomal curcumin formulations (including propylene glycol nanoliposomes and deformable liposome-chitosan hydrogels) achieved significantly higher drug concentrations in the skin with extended retention times compared to free curcumin. In one study, the liposomal format demonstrated early-stage healing properties in burn wound models that free curcumin could not match.
Growth factor stabilization. Growth factors like bFGF (basic fibroblast growth factor) and EGF (epidermal growth factor) are powerful wound healing promoters, but they degrade rapidly in conventional topical formats. The review highlights liposomes with silk fibroin hydrogel cores that provided dual slow-release properties for bFGF, nearly doubling wound healing rates compared to conventional liposome groups by day 14. A film-forming spray incorporating liposomes and chitosan achieved controlled release of EGF with uniform distribution across the wound surface.
Enhanced skin penetration. Several formulations in the review used penetration enhancers integrated directly into the liposome structure. Ethosomes (liposomes modified with ethanol) altered the lipid structure of the stratum corneum to improve permeation. Propylene glycol-modified liposomes significantly boosted drug concentrations and retention time on the skin. These approaches demonstrate that the liposome itself can be engineered as a penetration tool, not just a passive carrier.
Stimulus-responsive release. One of the more forward-looking sections of the review covers liposomes that release their payload in response to specific environmental triggers. Temperature-responsive liposomes (like the PECE-modified madecassoside liposome) transition from liquid to gel at body temperature, adhering to the wound surface and providing sustained drug delivery. pH-responsive liposomes accumulate preferentially at acidic wound sites. These “smart” delivery behaviors have clear implications for consumer topicals designed to respond to the skin’s own signals.
Clinical validation worth noting: The review documents several liposomal topical systems that have progressed to human clinical trials. A liposomal PVP-iodine hydrogel significantly accelerated healing in burn patients compared to silver sulfadiazine cream in a randomized controlled study of 43 patients. An insulin liposomal chitosan gel achieved a 16-fold increase in healing rate compared to control groups. These are consumer-relevant formats (gels, hydrogels, sprays) with human data behind them.
The research also reinforces something we have been seeing across supplement categories: targeted micronutrient strategies work better when paired with delivery systems that actually get the active ingredient where it needs to go. A topical recovery product with liposomal-encapsulated curcumin and a companion oral supplement with clinically dosed B vitamins creates a multi-pathway recovery system that conventional topicals simply cannot replicate.
Where Liposomal Topicals Fit on Your Product Roadmap
The clinical wound healing research reviewed in this paper is focused on medical applications, but the delivery technology, the active ingredients, and the consumer demand all translate directly into supplement and wellness product categories that are growing right now.
Recovery and muscle relief topicals. Cooling creams and muscle gels are one of the largest topical supplement categories, and most of them rely on the same basic formula: menthol or camphor for sensation, maybe some hemp extract or arnica for label appeal, and a standard cream base that deposits most of the actives on the skin surface. Liposomal encapsulation of functional ingredients like curcumin, hemp extract, or magnesium could meaningfully increase dermal penetration and provide sustained release over hours rather than minutes. That turns a surface-level sensation product into a genuine recovery tool.
Topical + ingestible recovery systems. The most compelling product strategy we see emerging from this research is the combination approach: a liposomal topical that delivers actives through the skin paired with an oral supplement (gummy, tincture, or recovery beverage) that supports the same recovery pathway from the inside. Topicals address local inflammation and tissue repair at the surface. Oral formats deliver systemic support for cellular repair and nutrient replenishment. Together, they create a recovery bundle with a clinical logic that consumers can understand and feel. As gummy formats continue to lead consumer adoption, pairing a recovery gummy with a liposomal topical gel makes for a cohesive product line with real market differentiation.
Skin wellness and anti-aging topicals. The review documents extensive work on liposomal systems carrying antioxidants (resveratrol, quercetin, anthocyanins) that protect against oxidative stress and promote skin regeneration. For wellness brands building clean-label skincare or beauty-from-within product lines, liposomal delivery of botanical antioxidants in a topical cream or serum offers a science-backed story that goes beyond conventional cosmetic formulation. The phospholipid content itself (typically from sunflower lecithin) contributes skin-conditioning properties, so the delivery system doubles as a functional ingredient.
Targeted functional tinctures. While the review focuses on topical wound healing, several of the liposomal systems described use the same phospholipid encapsulation technology that works in oral liquid supplements. A tincture with liposomal curcumin, liposomal glutathione, or liposomal B-complex vitamins delivers higher bioavailability through the GI tract using the exact same structural principle that makes liposomal topicals more effective at the skin level. Brands that understand the technology can apply it across both oral and topical product lines with a consistent scientific narrative.
The Competitive Window Is Open
Most consumer topical supplements on the market today are still using conventional delivery systems. The liposomal wound healing research reviewed in this paper has been progressing steadily in clinical and preclinical settings for years, but the consumer supplement market has been slow to adopt it. That gap between what the science can do and what most brands are actually offering represents a real opportunity for brands willing to invest in better formulation.
As AI-driven product discovery platforms continue to reshape how consumers evaluate supplements, brands with clinically grounded delivery stories will have a measurable advantage. The platforms reward transparency, specificity, and verifiable science. Liposomal topicals check all three boxes.
The research is there. The manufacturing technology is there. The consumer readiness is there. The question is whether your product line is ready to take advantage of it.
Ready to Explore Liposomal Topical Formulations?
OSM manufactures custom topicals, gummies, tinctures, and beverages with low MOQs and full turnkey services from our USDA Organic certified, GMP facility. Let’s build your next product around delivery science that works.
Start a ConversationSource: Ma, Z., Zhang, K., Luo, J., Chen, S., Tan, S. & Ma, D. (2026). “Liposome-based drug delivery systems for skin wound healing: a promising drug delivery strategy.” Frontiers in Bioengineering and Biotechnology, 14:1756872. doi: 10.3389/fbioe.2026.1756872
This article is for informational purposes only and does not constitute medical advice. The research reviewed focuses on clinical wound healing applications; supplement brands should consult regulatory counsel regarding product claims for consumer topical and oral products. Organic Supplement Manufacturing (OSM) is a contract manufacturer of dietary supplements, gummies, tinctures, beverages, and topicals operating from a USDA Organic certified, FDA-registered, third-party GMP certified facility in Plain, Wisconsin.

