OSM Industry Intel • Spring 2026
American Elderberry Juice Just Posted a Signal for Cognitive Flexibility in Mild Cognitive Impairment
A new randomized clinical trial in Antioxidants shows daily elderberry juice consumption improved cognitive flexibility speed in older adults with mild cognitive impairment after six months. Here is what supplement brands should be paying attention to.
Botanical ingredients with cognitive claims tend to fall into two buckets. Either the marketing gets way out ahead of the data, or the data exists but it is buried in animal studies and rat hippocampus assays that nobody outside a lab cares about. Neither one is great for a brand trying to build a supplement line that holds up under scrutiny.
Every once in a while though, a botanical actually puts up real numbers in real humans. American elderberry just did.
A randomized, double-blind, placebo-controlled clinical trial published in Antioxidants in January 2025 looked at what happens when older adults with mild cognitive impairment drink a small daily dose of American elderberry juice (Sambucus nigra subsp. canadensis) for six months. The headline finding: a statistically significant interaction between time and treatment on cognitive flexibility latency, with the elderberry group trending toward faster cognitive flexibility performance from baseline to six months, while the placebo group stayed flat.
That is the kind of result that should make a formulator pay attention. Not because elderberry is suddenly a cognition miracle (it is not, and the authors are explicit about that), but because the cognitive supplement category has been starving for botanicals with mechanistic backing AND human clinical signal at consumer-feasible doses. This study delivers both.
Here is the rundown on what the study actually found, why it matters for product development, and where elderberry fits into the broader 2026 cognitive supplement opportunity.
What the Elderberry Trial Actually Measured
The study, led by Musich and colleagues at the University of Missouri, was a secondary analysis of a registered clinical trial (NCT02414607). Twenty-four older adults (mean age 76) with a clinically confirmed mild cognitive impairment (MCI) diagnosis were randomized to either American elderberry juice (n=11) or a flavored-water placebo (n=13). Both groups consumed 5 mL of juice three times daily for six months.
The elderberry juice was commercially sourced from a Missouri grower and stabilized with citric acid, delivering 15.9 mg of cyanidin-3-glucoside per 5 mL, which is the primary anthocyanin in elderberry. That works out to roughly 48 mg of cyanidin-3-glucoside per day, which sits comfortably within doses already shown to be well tolerated in earlier human studies.
Participants completed a battery of cognitive tasks at baseline, three months, and six months: the Mini-Mental State Examination, the Hopkins Verbal Learning Test, the Rey-Osterrieth Complex Figure Test, an anagram-solving task, and a visuospatial problem-solving task. Rather than analyzing each task in isolation (which inflates Type 1 error and obscures small signals), the researchers built standardized z-score composites for four cognitive domains: global cognition, visuospatial construct ability, memory, and cognitive flexibility (accuracy, speed, and a combined rate-correct score).
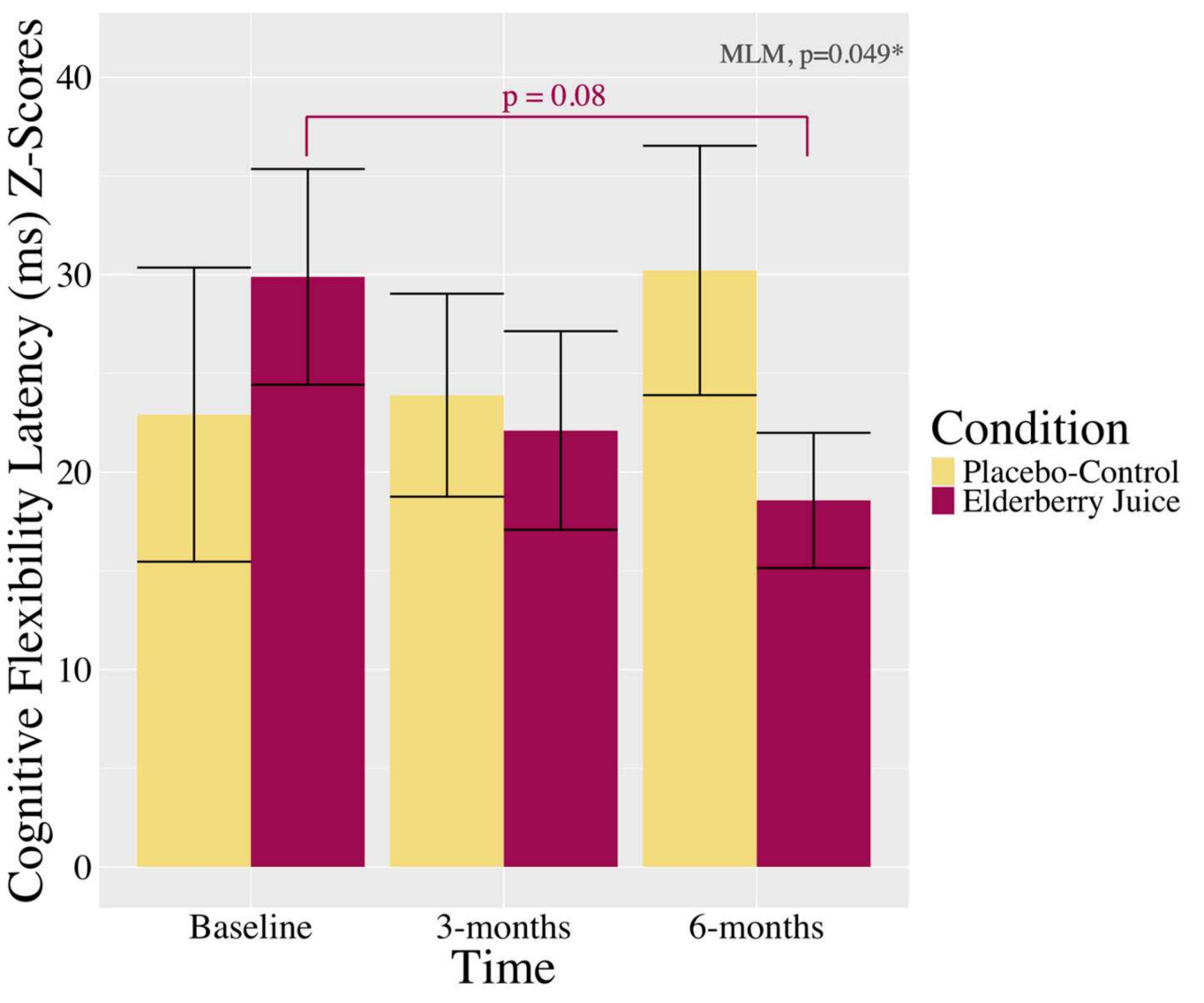
The Headline Result: Cognitive Flexibility Speed
The multilevel models surfaced one clean signal. The interaction between condition and time on the cognitive flexibility latency composite was statistically significant (p = 0.049, η² = 0.077, a medium effect size). Post-hoc pairwise comparisons showed the elderberry group’s cognitive flexibility latency dropped from a mean composite z-score of 29.89 at baseline to 18.57 at six months, a trend toward significance (p = 0.08) with a Hedge’s g of 0.72, which is a medium-to-large effect by Cohen’s conventions.
The placebo group did not budge. No meaningful change at any time point.
Lower latency means faster solution times on the anagram and visuospatial problem-solving tasks, which is to say the elderberry group got measurably quicker at solving novel cognitive flexibility problems over the course of six months. Accuracy did not change much in either group, but the authors note this is likely because participants were not making errors so much as running out of time. Latency is the more sensitive metric here.
THE FORMULATOR’S TAKE
A medium effect size on a clinical cognitive outcome, from a botanical, at a real-world consumable dose, over six months, in a population that is actively losing cognitive function. That is a much bigger deal than the small sample size makes it sound. The authors are appropriately cautious about overinterpreting a pilot trial, but the mechanism is plausible and the signal is in the right direction.
Why Anthocyanins, and Why Cognitive Flexibility Specifically
The mechanistic story here is not new, but the human evidence keeps stacking up. Anthocyanins (the pigments that make elderberries that deep purple-black) are among the most potent dietary antioxidants known, and a growing body of work shows they cross the blood-brain barrier in measurable quantities. Once there, they appear to modulate neuroinflammation, attenuate mitochondrial oxidative stress, and protect neural architecture in the frontal and parietal regions that govern executive function.
The companion paper from this research group (Curtis et al., 2024) showed that the same elderberry intervention reduced low-grade peripheral inflammatory markers in this MCI population. Inflammation is increasingly recognized as a driver of cognitive decline and Alzheimer’s pathology, so a botanical that dampens systemic inflammation AND shows a cognitive flexibility signal in the same population is doing what the mechanistic models predict it should do.
Cognitive flexibility (the ability to shift mental sets, solve novel problems, and adapt thinking to new constraints) sits squarely in the prefrontal cortex, which happens to be one of the regions where anthocyanin-mediated reductions in oxidative stress have been documented in animal models. The fact that this is the domain that moved, and not memory or general cognition, is consistent with the underlying biology rather than a random hit.
The Caveats Worth Knowing
This is a pilot trial with 24 participants. The effect on flexibility latency was statistically significant for the interaction but only trending in the pairwise comparison. There were no significant changes in memory, visuospatial construct ability, or global cognition composites. Family-wise error corrections were not applied. The sample was demographically homogeneous (all White, non-Hispanic, all from a single memory clinic).
All of that is fine for a pilot. It is also exactly the kind of finding that needs a larger, longer, more diverse follow-up trial before anybody starts making structure-function claims. From a brand and formulator standpoint, the takeaway is not “elderberry treats MCI.” The takeaway is that the cognitive support category has another evidence-backed botanical that fits into existing brand narratives around healthy aging, antioxidant support, and brain wellness.

Where Elderberry Fits in the 2026 Cognitive Supplement Stack
The cognitive support category has been one of the fastest-growing supplement segments for three years running, and it shows no signs of slowing down. Consumers over 55 are looking for botanicals they can recognize, pronounce, and feel good about taking daily. Elderberry checks every one of those boxes. It already has strong household recognition from the immune support category, which gives any new cognitive positioning a built-in trust runway that ingredients like bacopa or huperzine A simply do not have.
Here is how the clinical evidence translates into format strategy for brands working with a contract manufacturer.
Format 1: Functional Cognitive Beverages and Shots
The clinical trial itself used a 5 mL liquid format taken three times daily, which maps almost perfectly onto a small-volume daily shot or a once-daily ready-to-drink wellness beverage. Liquid elderberry preserves anthocyanin bioavailability better than powdered or encapsulated forms, and the natural sweetness and color are a formulation advantage rather than a problem to solve. Pair elderberry with complementary cognitive support ingredients like blueberry concentrate, B-complex, or lion’s mane and you have a category-leading SKU with a clinical hook on the lead ingredient.
Format 2: Cognitive Gummies for the Healthy Aging Consumer
Elderberry gummies are already a top-five seller in the immune category, and the manufacturing infrastructure is mature. The opportunity here is repositioning. Instead of competing in the saturated kids/family immune gummy space, brands can build a healthy-aging cognitive support line targeted at consumers 55+, with elderberry as the lead anthocyanin and supporting ingredients selected for synergistic mechanism (think omega-3 DHA, phosphatidylserine, or B6/B12). The clinical dose of 48 mg cyanidin-3-glucoside per day is achievable in a two-gummy serving with the right standardized elderberry extract.
For more on where the gummy category is heading and what brands need to be ready for in 2026, see our earlier breakdown of the four key imperatives for gummy manufacturing this year.
Format 3: Liposomal Elderberry Tinctures
Anthocyanins are notoriously fragile in the GI tract and have variable absorption depending on the food matrix they arrive in. A liposomal delivery system protects the active compound through digestion and improves systemic bioavailability, which matters more for cognitive endpoints than for immune endpoints because the active needs to reach the brain. A liposomal elderberry tincture is a premium positioning play that lets brands command higher margins while delivering on a real mechanistic advantage. We covered the technology in depth in our recent piece on how liposomal supplements work and where they fit in your product line.
Format 4: Stacked Cognitive + Recovery Formulations
The active adult and masters athlete demographic (think 50+ consumers who still train hard) is one of the most underserved segments in cognitive supplements. These buyers care about both physical recovery and mental sharpness, and they read labels carefully. An elderberry-based cognitive formula stacked with something like B3 and B6 for muscle recovery would land squarely in that white space. Worth reading our prior coverage of why B3 and B6 just got a lot more interesting for muscle recovery if you are thinking about combination products.
Compliance and Claims Reality Check
This is one clinical trial in a pilot population. It is not a basis for disease-treatment claims, and any brand bringing an elderberry cognitive product to market needs to stay firmly inside structure-function claim territory. Language like “supports cognitive flexibility,” “supports healthy brain aging,” and “antioxidant support for the brain” is defensible. Language that implies treatment, prevention, or reversal of MCI or Alzheimer’s is not, and FDA enforcement on cognitive claims has been notably more aggressive over the past 18 months. AI-driven supplement discovery tools are also surfacing these claim violations faster than ever, which we covered in our analysis of how AI is changing supplement discovery. Build your claim architecture conservatively and let the ingredient story do the heavy lifting.
The Bottom Line
Anthocyanin-rich botanicals have been promising on paper for two decades. The American elderberry data is one of the cleaner human signals to emerge in the cognitive support category in recent memory, and it lands at a moment when consumer demand for evidence-backed brain wellness products is at an all-time high. For brands building or expanding a cognitive line, elderberry is now a serious ingredient with serious data behind it. The formulation, sourcing, and claim architecture all matter, but the door is open.
Ready to Build?
Bring your elderberry cognitive product to market with a USDA Organic and GMP-certified manufacturer.
Organic Supplement Manufacturing handles formulation, sourcing, manufacturing, and fulfillment for gummies, tinctures, beverages, shots, and topicals. Low MOQs, full-service support, and a team that actually reads the clinical literature.
Start a Conversation →Source: Musich M, Curtis AF, Ferguson BJ, Drysdale D, Thomas AL, Greenlief CM, Shenker JI, Beversdorf DQ. Preliminary Effects of American Elderberry Juice on Cognitive Functioning in Mild Cognitive Impairment Patients: A Secondary Analysis of Cognitive Composite Scores in a Randomized Clinical Trial. Antioxidants. 2025; 14(2):131. https://doi.org/10.3390/antiox14020131
Disclaimer: This article is intended for supplement brand and contract manufacturing audiences for informational and educational purposes only. It is not intended as medical advice, and the findings discussed are preliminary. Statements regarding dietary supplements have not been evaluated by the FDA and are not intended to diagnose, treat, cure, or prevent any disease.

